Lithium-ion batteries have long been the standard for portable electronic devices and electric vehicles, providing a reliable source of energy for our modern, on-the-go lifestyles.
However, in recent years, a new contender has emerged in the world of energy storage – the Lithium Iron Phosphate (LiFePO4) battery.
With its distinct advantages and unique characteristics, the LiFePO4 battery has garnered significant attention and is poised to challenge the dominance of traditional lithium-ion batteries.

In this brief comparison, we will explore the key differences and advantages of Lithium iron phosphate battery vs. lithium-ion counterparts.
What is a Lithium Iron Phosphate Battery?
A Lithium Iron Phosphate (LiFePO4) battery is a specific type of lithium-ion battery that stands out due to its unique chemistry and components. At its core, the LiFePO4 battery comprises several key elements.
The cathode, which is the positive electrode, is composed of lithium iron phosphate (LiFePO4). This compound consists of lithium ions (Li+), iron (Fe), oxygen (O), and phosphate (PO4^3-) groups. This particular cathode material is chosen for its stability, safety, and performance characteristics. LiFePO4 has a robust crystal structure that is less prone to thermal runaway and overheating, making LiFePO4 batteries inherently safer than some other lithium-ion chemistries.
The anode, the negative electrode, is typically made of graphite, a common choice in lithium-ion batteries. Graphite stores lithium ions when the battery is charged and releases them during discharge.
An electrolyte, which is a conductive medium, enables the movement of lithium ions between the cathode and anode during the charge and discharge processes. It typically consists of a lithium salt dissolved in a solvent.
A separator is used to physically separate the cathode and anode while permitting the passage of lithium ions. This separator is crucial in preventing short circuits and maintaining the safety of the battery.
The chemistry of LiFePO4 batteries offers several advantages when comparing Lithium iron phosphate battery vs. lithium-ion batteries. These batteries are utilized in diverse applications, including electric vehicles, renewable energy storage systems, uninterruptible power supplies (UPS), and portable electronics, thanks to their stable voltage output, rapid charging capabilities, and safety profile.

Pros and Cons of Lithium Iron Phosphate Battery
Lithium Iron Phosphate (LiFePO4) batteries have several advantages and disadvantages, which make them suitable for certain applications but less ideal for others. Here are the key pros and cons of LiFePO4 batteries:
Pros:
- Safety:LiFePO4 batteries are renowned for their safety. They are less prone to thermal runaway, overheating, and the risk of fire or explosion compared to some other lithium-ion battery chemistries. This is due to the stable and robust crystal structure of LiFePO4.
- Long Cycle Life:LiFePO4 batteries offer a long cycle life, capable of enduring thousands of charge and discharge cycles without significant capacity loss when using a Lithium iron phosphate battery vs. lithium-ion battery. This makes them highly durable and cost-effective over the long term.
- Stability:LiFePO4 batteries maintain a relatively stable voltage throughout most of their discharge cycle. This characteristic ensures consistent power output, which can be crucial for applications requiring a steady power supply.
- Wide Temperature Range: LiFePO4 batteries can operate effectively in a wide range of temperatures, from extreme cold to high heat. This versatility makes them suitable for use in various environmental conditions.
- Fast Charging:LiFePO4 batteries can accept high charge and discharge currents, enabling rapid charging and discharging. This feature is advantageous in applications where quick power delivery is essential.
- Environmental Friendliness: LiFePO4 chemistry is considered environmentally friendly as it does not contain toxic heavy metals like cobalt or nickel. It is a greener option compared to some other lithium-ion chemistries.
- Low Self-Discharge:LiFePO4 batteries have a lower self-discharge rate when comparing Lithium iron phosphate battery vs. lithium-ion counterparts compared to some other battery types, which means they can hold their charge for longer periods when not in use.
Cons:
- Higher Cost:LiFePO4 batteries can be more expensive upfront compared to some other lithium-ion batteries due to the cost of the raw materials and manufacturing processes. However, their long cycle life can offset this initial cost in certain applications.
- Lower Energy Density:LiFePO4 batteries typically have a slightly lower energy density compared to some other lithium-ion chemistries. This means they store less energy per unit of weight, which can limit their use in applications where space and weight constraints are critical.
- Bulkier and Heavier:Because of their lower energy density, LiFePO4 batteries may be bulkier and heavier for a given energy capacity compared to batteries with higher energy densities. This can impact their suitability for portable applications.
- Complex Battery Management:LiFePO4 batteries may require more complex battery management systems to ensure proper charging and discharging to maximize their lifespan and safety.
What is a Lithium-ion battery?
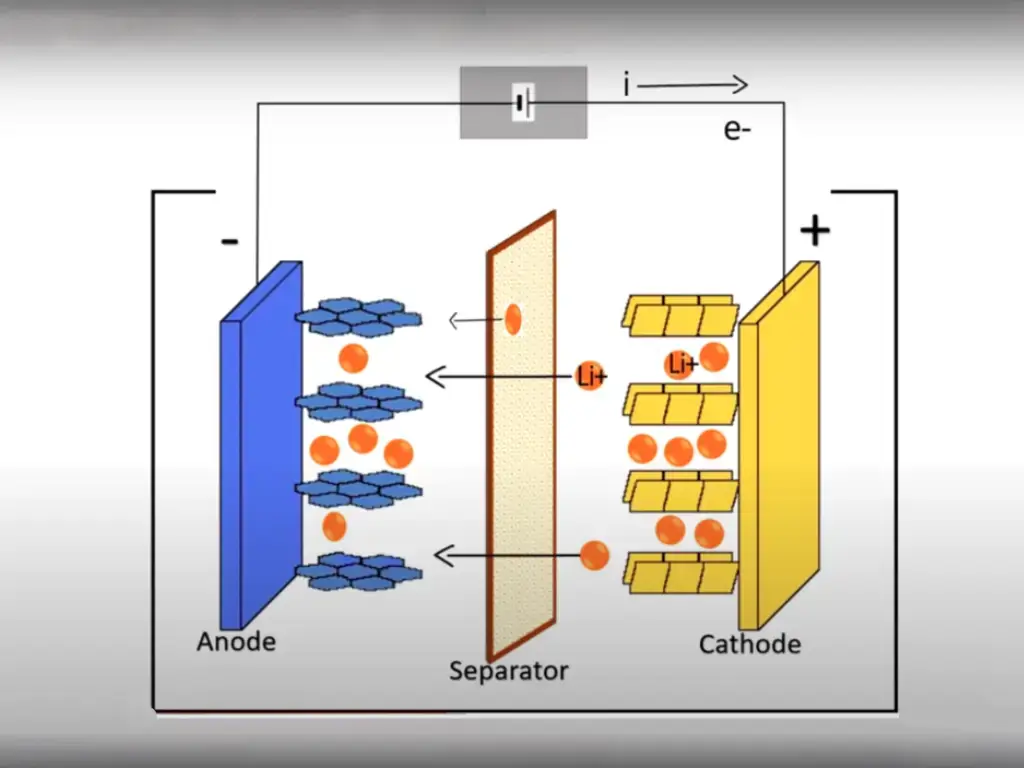
A Lithium-ion (Li-ion) battery is a rechargeable energy storage device that relies on the movement of lithium ions between the battery’s positive and negative electrodes to store and release electrical energy.

The fundamental chemistry behind Li-ion batteries involves the use of lithium as the primary charge carrier. In a Li-ion battery, the negative electrode, or anode, typically consists of a form of carbon (such as graphite), which can intercalate or absorb lithium ions during charging. The positive electrode, or cathode, is typically made of a lithium metal oxide compound, which can release lithium ions during discharge.
These two electrodes are separated by an electrolyte, which is typically a lithium salt dissolved in a solvent. The movement of lithium ions between the electrodes during charging and discharging is facilitated by the electrolyte, and this flow of ions generates the electrical current that powers electronic devices.
Li-ion batteries such as 12V lithium-ion golf batteries are prized for their high energy density, making them compact and lightweight while offering a reliable and rechargeable power source for various applications. From smartphones and laptops to electric vehicles and renewable energy storage systems these batteries serve as an essential part of these system. Moreover, 12V lithium-ion golf batteries
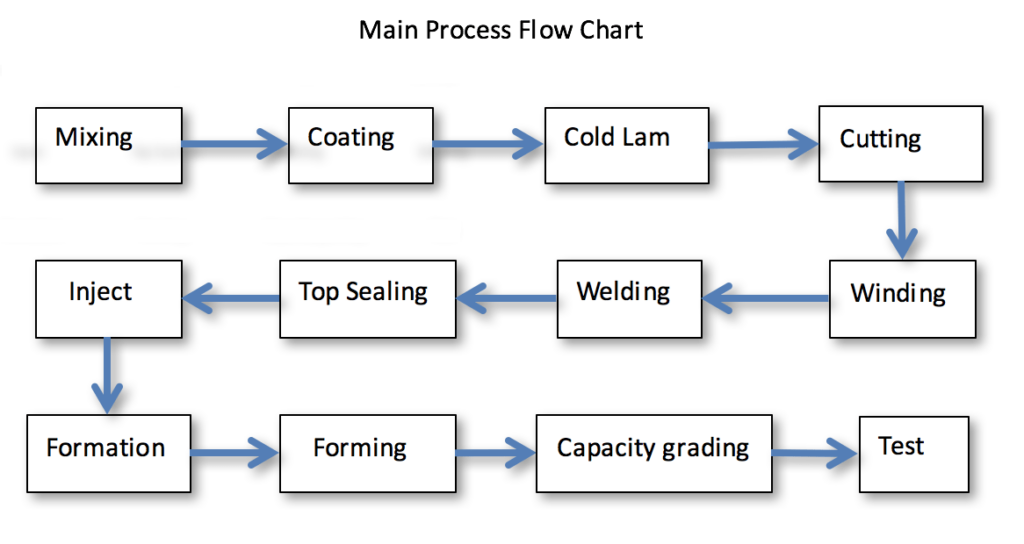
Below is the lithium-ion battery manufacturing flow chart:
Pros:
- High Energy Density:Li-ion batteries offer a high energy density when comparing Lithium iron phosphate battery vs. lithium-ion, which means they can store a significant amount of energy relative to their size and weight. This makes them ideal for portable electronic devices like smartphones, laptops, and tablets.
- Rechargeable: Li-ion batteries are rechargeable, allowing them to be used multiple times before needing replacement. This reusability reduces waste and is cost-effective over the long term.
- Low Self-Discharge:Li-ion batteries have a relatively low self-discharge rate, meaning they can hold their charge for extended periods when not in use. This makes them suitable for applications where sporadic use is common.
- No Memory Effect: Li-ion batteries do not suffer from the memory effect, a phenomenon that can reduce the capacity of some rechargeable batteries if they are not fully discharged before recharging. Users can charge Li-ion batteries at any state of charge without harming their performance.
- Fast Charging:Li-ion batteries can be charged quickly, which is beneficial for devices that require rapid replenishment of power.
Cons:
- Safety Concerns:Li-ion batteries can pose safety risks if overcharged, punctured, damaged, or exposed to high temperatures when comparing Lithium iron phosphate battery vs. lithium-ion There have been incidents of Li-ion batteries catching fire or exploding, although safety measures and improvements have reduced these risks.
- Environmental Impact:The production and disposal of Li-ion batteries can have environmental consequences due to the extraction and processing of lithium, cobalt, and other raw materials. Efforts are ongoing to make Li-ion batteries more sustainable.
- Cost: Li-ion batteries can be expensive to manufacture, primarily due to the cost of raw materials like lithium and cobalt. However, economies of scale and advancements in technology have been gradually reducing costs.
- Heat Sensitivity: Li-ion batteries are sensitive to temperature extremes. Operating them in very high or low temperatures can affect their performance and lifespan.
- Capacity Fade:Over time, Li-ion batteries experience capacity fade, where their ability to hold a charge diminishes. This can lead to reduced runtimes for devices and the need for more frequent recharging.
Lithium Iron Phosphate Battery Vs. Lithium-Ion: Comparing The Two Batteries

Here’s a comparison table for Lithium iron phosphate battery vs. lithium-ion:
|
Aspect
|
Lithium Iron Phosphate (LiFePO4)
|
Lithium-ion (Li-ion)
|
|---|---|---|
|
Chemical CompositiCoston
|
Uses lithium iron phosphate (LiFePO4) as the cathode material.
|
Utilizes various lithium metal oxides in non-universal lithium-ion batteries, such as lithium cobalt oxide (LiCoO2) or lithium manganese oxide (LiMn2O4), as cathode materials.
|
|
Size and Weight
|
It may be bulkier and heavier for a given energy capacity due to lower energy density.
|
They are compact and lightweight, making them suitable for portable electronics and mobile devices.
|
|
Cycle Life
|
Longer cycle life, often exceeding 2,000-3,000 cycles with minimal capacity loss.
|
Good cycle life, typically 300-500 cycles, but can vary based on chemistry and usage.
|
|
Energy Density
|
Slightly lower energy density when comparing Lithium iron phosphate battery vs. lithium-ion, providing less energy per unit weight.
|
Higher energy density, offering more energy in a lighter package
|
|
Charge/Discharge Rate
|
Capable of accepting high charge and discharge currents, enabling rapid charging and discharging.
|
Good charge/discharge rates, but may not be as fast as LiFePO4 in some cases.
|
|
Temperature Range
|
Operates effectively in a wide temperature range, from -20°C to 60°C or more
|
Requires more controlled temperature conditions for optimal performance and safety.
|
|
Voltage Stability
|
Offers relatively stable voltage output throughout most of the discharge cycle.
|
Voltage output tends to decrease linearly during discharge.
|
|
Safety
|
Excellent safety profile with low risk of thermal runaway or fire.
|
Good safety but with a slightly higher risk of thermal issues compared to LiFePO4. Also, there can be lithium-ion battery leaking seen, which can be dangerous.
|
|
Applications
|
Ideal for applications where safety, longevity, and stability are critical, such as electric vehicles, renewable energy storage, and critical backup systems.
|
Widely used in portable electronics, laptops, smartphones, and many consumer devices where energy density and weight are key
|
|
Cost
|
Typically, higher upfront costs due to the cost of raw materials and manufacturing.
|
When comparing Lithium iron phosphate battery vs. lithium-ion, It may have a lower initial cost, but the overall cost of ownership can vary based on factors like cycle life.
|
Lithium iron phosphate battery vs. lithium-ion: Which One Is The Best?
In the comparison between Lithium iron phosphate battery vs. lithium-ion there is no definitive “best” option. Instead, the choice should be driven by the particular demands of the application. LiFePO4 batteries excel in safety, longevity, and stability, making them ideal for critical systems like electric vehicles and renewable energy storage.
In contrast, Li-ion batteries offer higher energy density, making them perfect for compact, lightweight, portable devices such as smartphones and laptops.
The selection should be a deliberate process, evaluating the specific requirements, safety concerns, energy needs, cost considerations, and environmental impact to determine which battery chemistry best aligns with the intended purpose. Ultimately, both LiFePO4 and Li-ion batteries have their roles to play in powering the diverse array of modern technologies.