Sodium ions are garnering attention worldwide, as an alternative to Lithium ions, in rechargeable batteries. Sodium ions are excellent charge carriers, as you can see from this simple Sodium Chloride and Water battery experiment.
Global leaders in the battery industry are placing big bets on Sodium-ion batteries as the future backbone of green industrial energy. If you look-up the world’s top 10 Sodium-ion battery manufacturers, you will notice that they also include the biggest manufacturers of Lithium-ion batteries.
Since Sodium ion and Lithium-ion battery chemistries are similar, manufacturers are leveraging proven Lithium-ion know how and production equipment for rapid development of Sodium-ion batteries. A good example is China’s Contemporary Amperex Technology Co. Ltd. (CATL), who are one of top10 Lithium-ion batteries manufacturers in China. CATL have launched their first generation Sodium-ion battery with energy density of 160 Wh/kg.
Since Sodium-ion batteries are bulkier than equivalent Lithium-ion batteries, they are not ideal for electric vehicles at present. Hence manufacturers are targeting the energy storage market, where size is not important but cost, performance and secure supply chains are key considerations. Scientists believe that Sodium-ion batteries are the answer to the renewable energy storage challenge.
In future, you can expect Sodium-ion batteries to become lighter and more powerful. This means that these batteries will become attractive for the electric vehicle market. However, you might have noticed that recently the electric vehicle market has not been robust. Globally, the market for electric vehicles has faltered, due to their high cost, lack of recharging infrastructure, and in many cases, withdrawal of fiscal incentives. Further, Lithium-ion batteries may be displaced by Hydrogen Fuel cells in the transportation sector. With Lithium-ion restricted to smaller electronic equipment, Sodium-ion technology can forge ahead in the long-term.
What’s The Sodium Ion Battery?
A Sodium ion battery is a rechargeable battery that uses Sodium ions to transport electric charge within the battery. Sodium ions shuttle between the Anode and Cathode, carrying the internal cell current, during each charge and discharge cycle.
How Does a Sodium Ion battery Work?
You can see the principle underlying a Sodium-ion battery in Figure 1. The key working parts of the battery are:
- An Anode, which is a porous electrode and contains plenty of ionizable Sodium atoms on its surface;
- A Cathode, which is also a porous electrode, with sufficient surface area to receive Sodium ions coming from the Anode;
- A porous separator that prevents physical contact between Anode and Cathode, but allows Sodium ions to shuttle between Anode and Cathode;
- A liquid electrolyte containing dissolved Sodium salts. The electrolyte acts as reservoir of Sodium ions that start an internal current once a voltage difference is created between the electrodes.
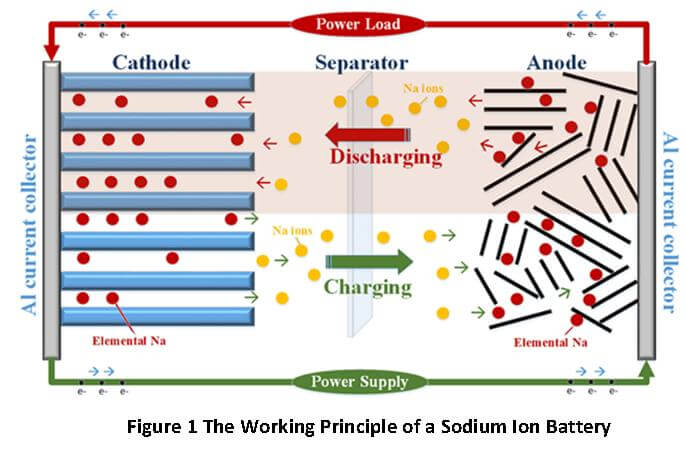
In Figure 1, the yellow balls represent positively charged Sodium ions while the red balls represent neutral Sodium atoms. Each neutral Sodium atom becomes a positively charged ion, when an electron is pulled out from its outermost electron shell.
So, once an external voltage is applied through a charger or a larger battery, electrons get pulled out from the Sodium atoms attached to the Anode. These electrons flow into the external circuit, and reach the Cathode. The Cathode acquires negative potential due to the accumulation of electrons.
Simultaneously, the Sodium atoms at the Anode, having lost one electron each, become positive ions and leave the Anode. Due to the potential difference, they travel through the electrolyte, towards the Cathode. At the Cathode, the positive Sodium ions are neutralized by electrons reaching there from the external circuit. The resultant neutral Sodium atoms park themselves at the Cathode during the remaining part of the charging cycle.
When an external load is connected and the battery discharges, the reverse journey starts for the Sodium ions parked at the Cathode. The Sodium ions travel back to the Anode through the electrolyte, while electrons from the Cathode travel through the external load to the Anode. Once at the Anode, Sodium-ions get neutralized by electrons coming in from the external circuit. The neutral Sodium atoms park themselves at the Anode during the rest of the discharge cycle. Thus, the entire life of Sodium ions in this type of rechargeable battery is spent in shuttling between the Anode and Cathode!
The maximum traffic density of Sodium ions depends upon the properties of the electrolyte as well as the Anode and Cathode construction. These are important parameters in battery design, determining the energy density and current.
Applications
Experts believe that the ideal applications for Sodium ion batteries are in stationary energy storage applications in the renewables and industrial sectors. The NAIMA project in the European union is focused on developing two configurations of eco-friendly, safe, Sodium-ion cells for the energy storage sector.
Meanwhile, the world’s first commercial deployment of Sodium-ion batteries in the automotive sector has been accomplished in China. A joint venture between JAC of China and Volkswagen of Germany launched a five-seater passenger vehicle powered by a 25 kwh Sodium ion battery, with a range of 250 km.
Sodium-ion batteries are also being considered for personal electronic devices, such as such as laptops and mobile phones. Though heavier than Lithium-ion batteries, they are cheaper and safer, both of which are important consumer considerations.
Another application of Sodium-ion batteries is in back-up power systems, such as in Natron Energy’s UPS .
How Sodium Ion Battery Changes The Game:
Sodium-ion batteries will bring many benefits into rechargeable battery markets. These include:
Higher Safety
Compared to Lithium-ion batteries Sodium-ion batteries are less prone to catch fire. Future generations of Sodium-ion may use solid state electrolytes which will be absolutely free from this hazard. For example, Australian researchers have developed a non-flammable solid polymer electrolyte, for use with Sodium-ion batteries. Various other strategies to mitigate thermal risks of Sodium-ion batteries are also being investigated including materials selection for improved safety.
Lower Cost
The cost of Sodium-ion batteries per kwh is less than one-third that of Lithium-ion batteries. Because of the raw material cost.
Lower Supply Chain Risk
Unlike Lithium, which is mined and refined only in a few countries, Sodium is easily available throughout the world. This reduces the raw material supply chain risks significantly.
Sodium Ion Battery Vs Lithium Ion Battery
As a consumer, or investor, if you had to make a choice between the two, which way would you go? You would look at immediate benefits and performance features as a consumer, whereas, as an investor you would look at larger, long-term issues.
Let us compare the two types of batteries across a few key parameters, relevant to consumers as well as investors.
| Parameter | Lithium-Ion battery | Sodium-Ion Battery |
| Maturity of technology and supply chains | Technology is mature, there are established global supply chains. | First generation technology is commercialized. Limited supply chains. |
| Cost | ~ $130 -$180/ kwh | ~ $40-$55/kwh |
| Safety | High risk of thermal runaway due to cell materials catching fire from uncontrolled heat production | Thermal runaway risk exists for Sodium ion batteries, but can be eliminated by cell materials selection and design in future. |
| Energy Density | 150 -250 Wh/kg | 100-160 Wh/kg |
| Voltage | 3.0 ~4.5 V | 2.8 ~3.5 V |
| Storage at zero charge | Not recommended, will reduce battery life | Permitted. Will not affect battery life |
| Supply Chain Risk | High, due to Lithium supply an refining concentrated in a few countries. | Low risk, since Sodium is abundantly available and refining is easy. |
Sodium Ion Batteries-Present And Future
Sodium-Ion batteries have made their entry at the right time, when global attention is focused on decarbonization of energy. The first-generation Sodium-ion batteries have already demonstrated several advantages over Lithium-ion, particularly on cost and safety parameters.
However, the current demand for such rechargeable batteries is driven by electric vehicles and personal electronic devices, where Lithium batteries dominate the market. Due to lower power density and more weight, it will be difficult for Sodium-ion to compete using first generation batteries. Hence a lot of research and development effort is going into improvement of the power density of Sodium-ion batteries. In fact a breakthrough may be near, since CATL announced that its next generation Sodium-ion battery will achieve 200 kwh/kg. Energy density improvement at competitive costs, will be a never-ending challenge for all types of batteries. For example CATL are again setting the pace here by working on a futuristic Lithium based 500 kwh/kg condensed matter battery.
In the medium term, increasing renewable energy investments by countries around the world will boost up the demand for energy storage systems. Sodium-ion battery manufacturers see this as the best option, once it takes-off.
Apart from energy and power density, Sodium-ion battery technology continues to develop, to close the gap with Lithium-ion technology. Scientists are resolving issues related to material degradation, developing cheaper and better anodes, cathodes and electrolytes, improving safety and recycle ability.
The supply chains for Sodium-ion batteries also have to expand, for which the necessary investments must be made.
In conclusion, Sodium ion batteries have emerged as a serious competitor to their Lithium-ion counterparts. However, it will take many years of development and commercial success before one can say that Sodium-ion batteries represent the future of energy.